Search
The Wesfarmers Centre is pleased to announce the successful applications for the 2016 Round 2 Wesfarmers Centre Seed Funding.
Eight applicants were successful, and were awarded $15,000 each for activities supporting subsequent research grant applications.
In 2014, the Wesfarmers Centre of Vaccines & Infectious Diseases put out a first call for seed funding proposals.

Preventing over half of the world’s ear infections with a therapy such as Spritz-OM will significantly improve health and educational outcomes on a global scale.

The Wesfarmers Centre has established the Deborah Lehmann Research Award to acknowledge the significant contribution that Clinical Associate Professor Deborah Lehmann AO has made to paediatric infectious disease research.

Dr Janessa Pickering is a research microbiologist with expertise in the molecular diagnostics and host pathogen interactions of upper respiratory tract pathogens that cause disease in children.

Program Manager
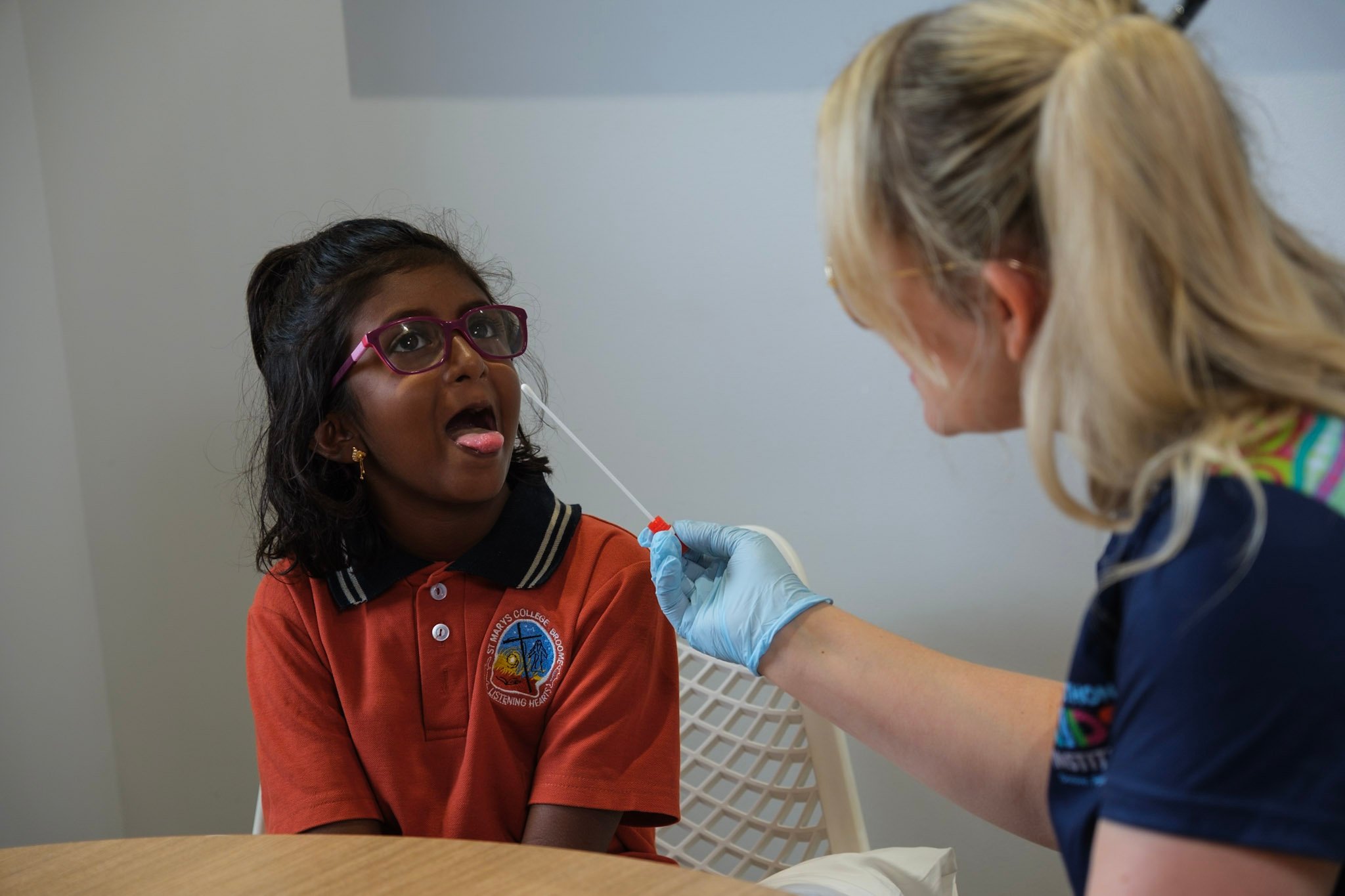
An alarming number of Strep A infections are going unnoticed throughout classrooms in Broome and Derby according to a major study by The Kids Research Institute Australia aiming to reduce the burden of acute rheumatic fever (ARF) and rheumatic heart disease (RHD).

Valuable support from the Raine Medical Research Foundation’s 2025 grant round will power four new research projects at The Kids Research Institute Australia.

Efforts to improve health outcomes for Aboriginal children have been accelerated thanks to almost $1 million in National Health and Medical Research Council (NHMRC) funds awarded to skin health researchers at The Kids Research Institute Australia.
