Search
Christopher Jennifer Peter Blyth Kent Richmond MBBS (Hons) DCH FRACP FRCPA PhD RN MBBS MRCP(UK) FRACP Centre Head, Wesfarmers Centre of Vaccines and
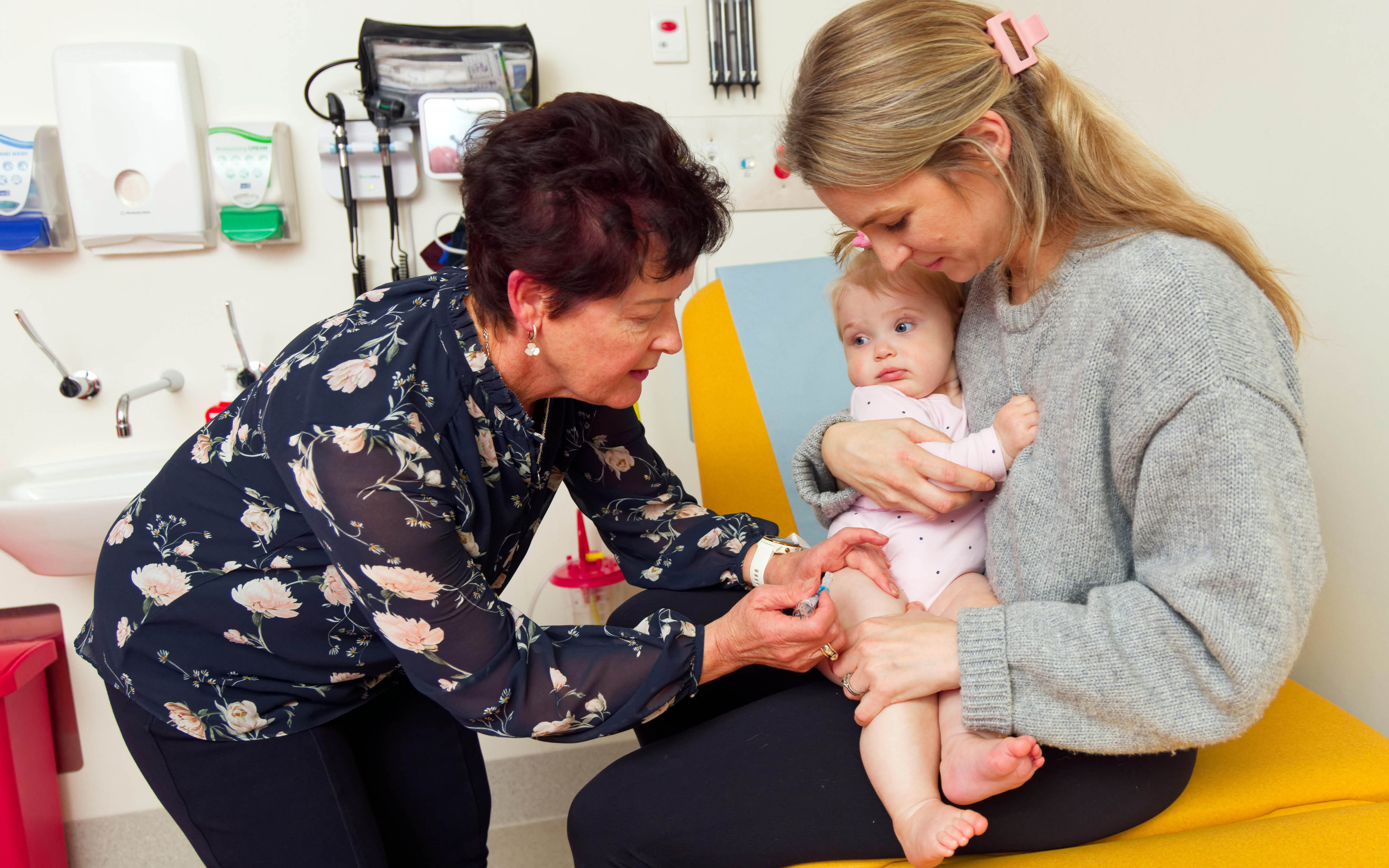
The mission of the Vaccine Trials Group is to improve the health of the community through immunisation and the prevention of infectious diseases.
B-cell epitope mapping is an approach that can identify and characterise specific antigen binding sites of B-cell receptors and secreted antibodies. The ability to determine the antigenic clusters of amino acids bound by B-cell clones provides unprecedented detail that will aid in developing novel and effective vaccine targets and therapeutic antibodies for various diseases.
The first few days of life are characterized by rapid external and internal changes that require substantial immune system adaptations. Despite growing evidence of the impact of this period on lifelong immune health, this period remains largely uncharted.
Early neurological deterioration (END) within 72 h of stroke onset is associated with poor prognosis. Optimizing hydration might reduce the risk of END.
Acute respiratory infections (ARI) are the most common cause of paediatric hospitalisation. There is an urgent need to address ongoing critical knowledge gaps in ARI management. The Pragmatic Adaptive Trial for Respiratory Infections in Children (PATRIC) Clinical Registry will evaluate current treatments and outcomes for ARI in a variety of paediatric patient groups.
The purpose of this study is to explore the effectiveness of a hospital-based asynchronous ear, nose, and throat telehealth service (the Ear Portal) in reducing cost and improving access for children with otitis media.
One third of children require repeat ventilation tube insertion (VTI) for otitis media. Disease recurrence is associated with persistent middle ear bacterial biofilms. With demonstration that Dornase alfa (a DNase) disrupts middle ear effusion biofilms ex vivo, we identified potential for this as an anti-biofilm therapy to prevent repeat VTI. First, safety and tolerability needed to be measured.
Varicella (chickenpox) is an infectious disease caused by the varicella zoster virus affecting children, adolescents, and adults. Varicella symptoms are usually self-limiting; however, different complications with widespread and systemic manifestations can occur. This systematic literature review aims to explore and quantify varicella-associated complication rates.
Pneumonia is the leading cause of death in young children globally and is prevalent in the Papua New Guinea highlands. We investigated clinical predictors of hypoxic pneumonia to inform local treatment guidelines in this resource-limited setting.
